|
VersaCross® Radiofrequency System Reduces Time to Left Atrial Access versus Conventional Mechanical Needle study led by Dr. Jacqueline Saw
Study highlights using RF wire:
|
|
|
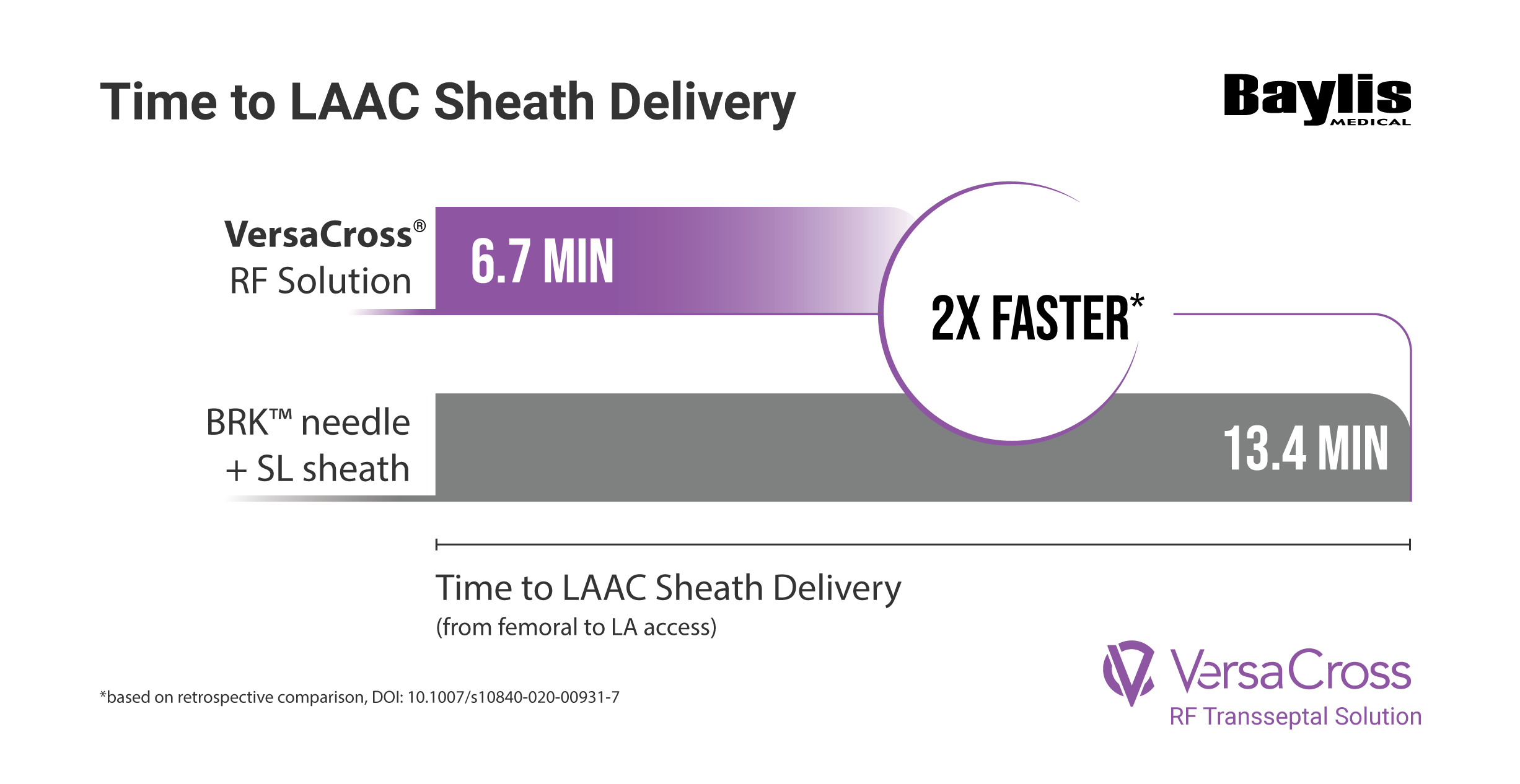
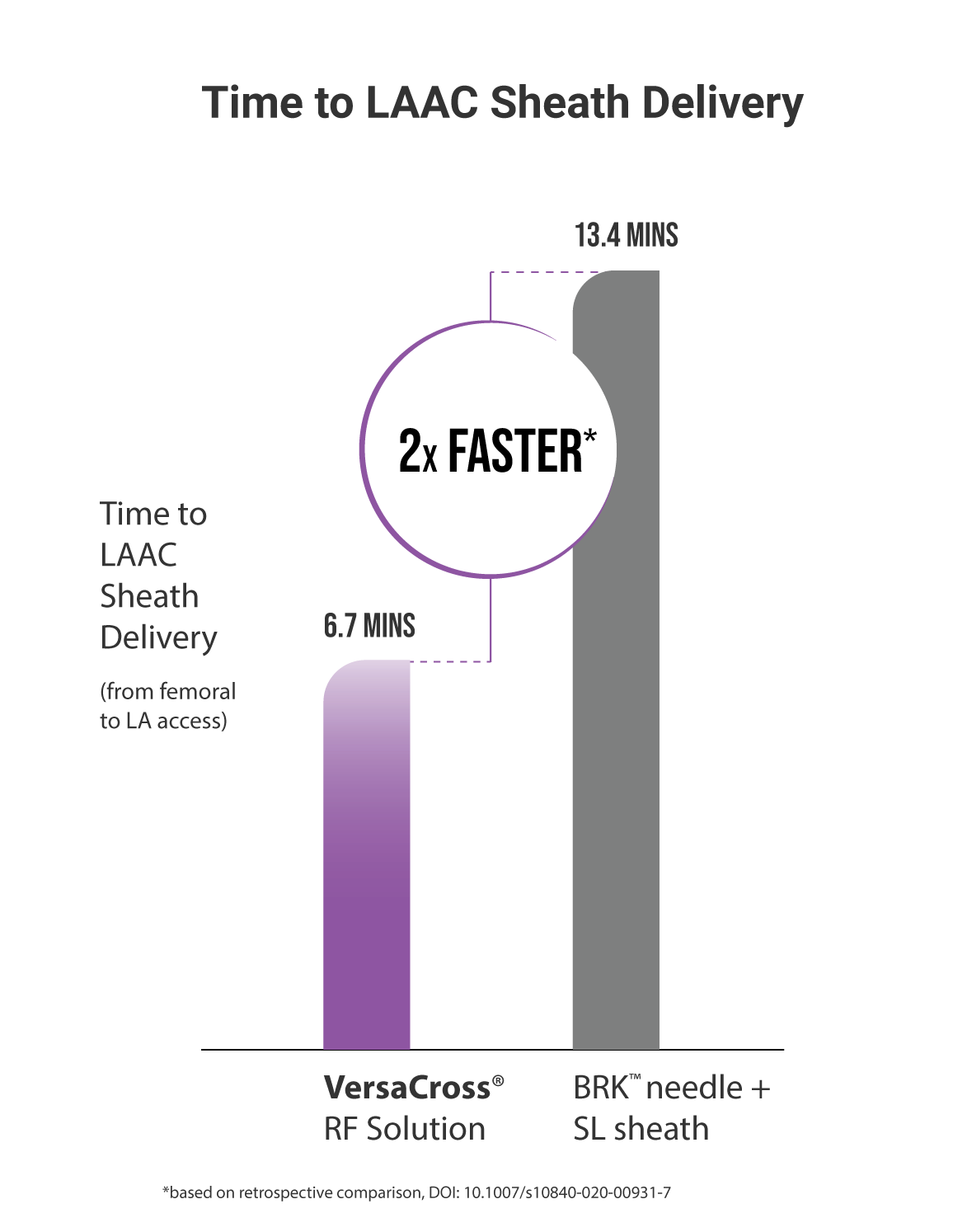
EFFICIENT: Achieved transseptal and Watchman™ or Amulet™ sheath delivery in under 7 mins
EXCHANGELESS: Enabled faster LA access with 3-in-1 solution by combining a starter wire, RF transseptal device and exchange rail
EFFORTLESS: Controlled RF puncture with a single wire
|
|
|
Watch Dr. Jacqueline Saw speak on this study
 |

Single Solution. No Exchanges.™
World’s first exchangeless solution for access-to-delivery of left heart therapy devices.
|
†compared to 13.4 mins using BRK needle in a retrospective comparison, VersaCross® RF Transseptal Solution delivered LAAC sheath in 6.7 mins, Inohara et al. J Interv Card Electrophys, 2021 DOI: 10.1007/s10840-020-00931-7
VersaCross® RF Transseptal Solution is intended for left heart access. The VersaCross® RF Wire is indicated for creation of an atrial septal defect in the heart. VersaCross® Transseptal Sheath is indicated for introducing cardiovascular catheters and guidewires to the left atrium.
PRM-00673 EN J-1,2,3 V-4 © Copyright Baylis Medical Company Inc., 2021. Baylis Medical Company Inc. reserves the right to change specifications or to incorporate design changes without notice and without incurring any obligation relating to equipment previously manufactured or delivered. VersaCross, the VersaCross logo, ‘Single Solution. No Exchanges.’, Baylis Medical and the Baylis Medical logo are trademarks and/or registered trademarks of Baylis Medical Company Inc. in the USA and/or other countries. Other trademarks are property of their respective owners. Patents Pending and/or issued. CAUTION: Federal Law (USA) restricts the use of these devices to or by the order of a physician. Before use, consult product labels and Instructions for Use for Indications for Use, Contraindications, Warnings, Precautions, Adverse Events and Directions for Use. Products shown may not be approved in all jurisdictions.
Thank you for subscription!